Colour coded chemicals
A team led by researchers at the La Trobe Institute for Molecular Sciences (LIMS) in collaboration with RMIT has been investigating a novel way to analyse chemicals using visible light.
The device uses the interaction between photons and electrons to filter out colours to create an ‘optical barcode’ that is specific to a sample and can be compared to a library of known material signatures.
The group’s tuneable and potentially portable device can quickly help to identify concentrations of chemicals and can be used for a range of sensing applications including monitoring water pollutants and inspecting soil quality. Because the device does not chemically modify the solution being analysed it has the potential to be used for real-time monitoring in applications including food and pharmaceutical production.
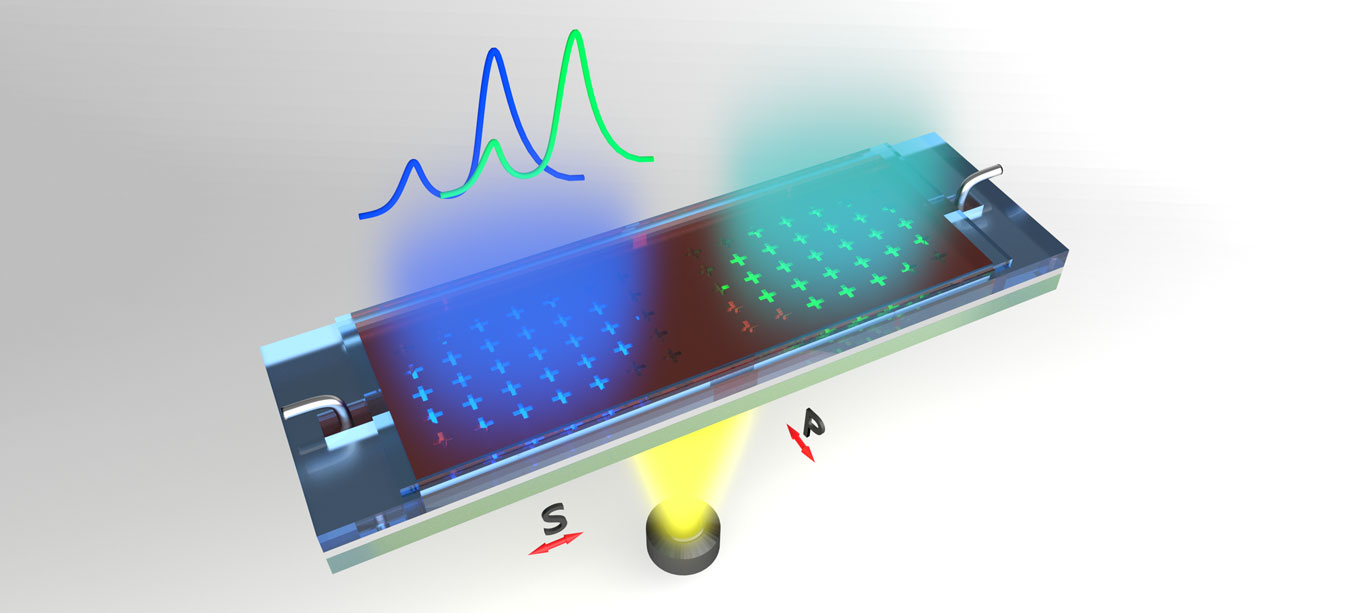
Using microfluidic channels, the device directs a liquid sample over a quartz surface with a silver coating. The coating features an array of nanometre-sized holes which allow light to pass through the device. To identify a sample, broadband light is passed through the liquid and is observed from the other side. Due to a phenomenon described by a branch of physics called plasmonics, all but one colour of light is filtered out as it passes through the liquid and the array depending on the refractive index of the sample.
The refractive index of a material is an intrinsic property that defines how fast light can pass through it. This refractive index can be used to help differentiate chemicals, so comparing the wavelength of the transmitted light to a known library can be used to help identify a sample.
This form of chemical sensing has been tested before, but it has run into problems detecting the minute changes in colour that occur for different chemicals.
The issue has now been overcome, thanks to the LIMS/RMIT team’s breakthrough which came by changing the device to be sensitive to not just the refractive index of the sample, but to the incident orientation of the light (polarisation) as well. This introduces a way to create a chain of reference points that can be used to identify chemicals far more accurately than ever before using this technique.
Plasmonic filtering relies on the fact that within the thin film of silver there is a “sea” of free elections. When the broadband light hits the surface, a particular wavelength of light resonates with these electrons depending on the distance between the holes in the coating and the refractive index of the sample to create a standing wave. Any light of this wavelength is allowed to pass through the holes, while all other light is absorbed or reflected by the silver layer. Historically, this type of sensing uses an evenly spread array of circular holes which means that it doesn’t matter what polarisation of light hits it.
The team instead created an array that features cross-shaped holes, and then spread them at different distances in the x and y planes. This means that the wavelength of light needed to excite the electrons between the crosses in the x-direction is different to the one needed to excite the electrons in the y-direction.
Using light that was polarised to align with either the x or y directions therefore produces different colours once the plasmonic filtering has occurred and effectively creates two reference points to compare to other materials. By testing a range of different angles of polarised light for the same sample and recording the series of colours produced then creates a far more specific signature to be compared to a library of known results.
The team used MCN’s Electron Beam Evaporation equipment to deposit the silver coating, before using the Centre’s Focused Ion Beam Scanning Electron Microscope (FIB-SEM) to mill out the nanometre-sized crosses.
“Data collection was also carried out at MCN using the microspectroscope equipment. Every step of the process from producing the devices to analysing the data involved support and collaboration with MCN,” said Dr Daniel Langley, who was the first author of the study.
The team now plans to use this technology to develop a standalone chemical sensor for probing local environments at the microscale in a variety of applications. Initially though, the aim is to tailor the device to agricultural applications, monitoring contamination in plants and soils.
“We would like to improve the sensitivity of the technique even further, refining the device structure and exploring different materials in the active layers.” Daniel said. “These developments will require close interaction with ANFF staff as we work out the possibilities at MCN.”
“We would also like to develop a library of ‘optical barcodes’ for different chemicals and mixtures – here ANFF’s broad network could be essential for making the research and industry connections we need to direct this technology towards the highest impact applications with the greatest societal benefit,” said A/Prof Brian Abbey, a co-corresponding author on the paper.