Diamond coating Carbon Fibre
Exploring gold nanowires and nanorods
Professor Wenlong Cheng from Monash University has been exploring unique properties of gold nanomaterials for over 16 years. Two recent projects, conducted at ANFF-Vic’s Melbourne Centre for Nanofabrication (MCN), involved gold nanorods and gold nanowires.
Wearable sensors
Stretchable, conductive materials are driving the capabilities of tomorrow’s soft and wearable technologies.
Wearables may hold the key to accurate and comfortable health monitoring and movement detection. There’s also the potential for a range of intuitive human/machine interfaces to be created that could ultimately enable implantable biomedical devices.
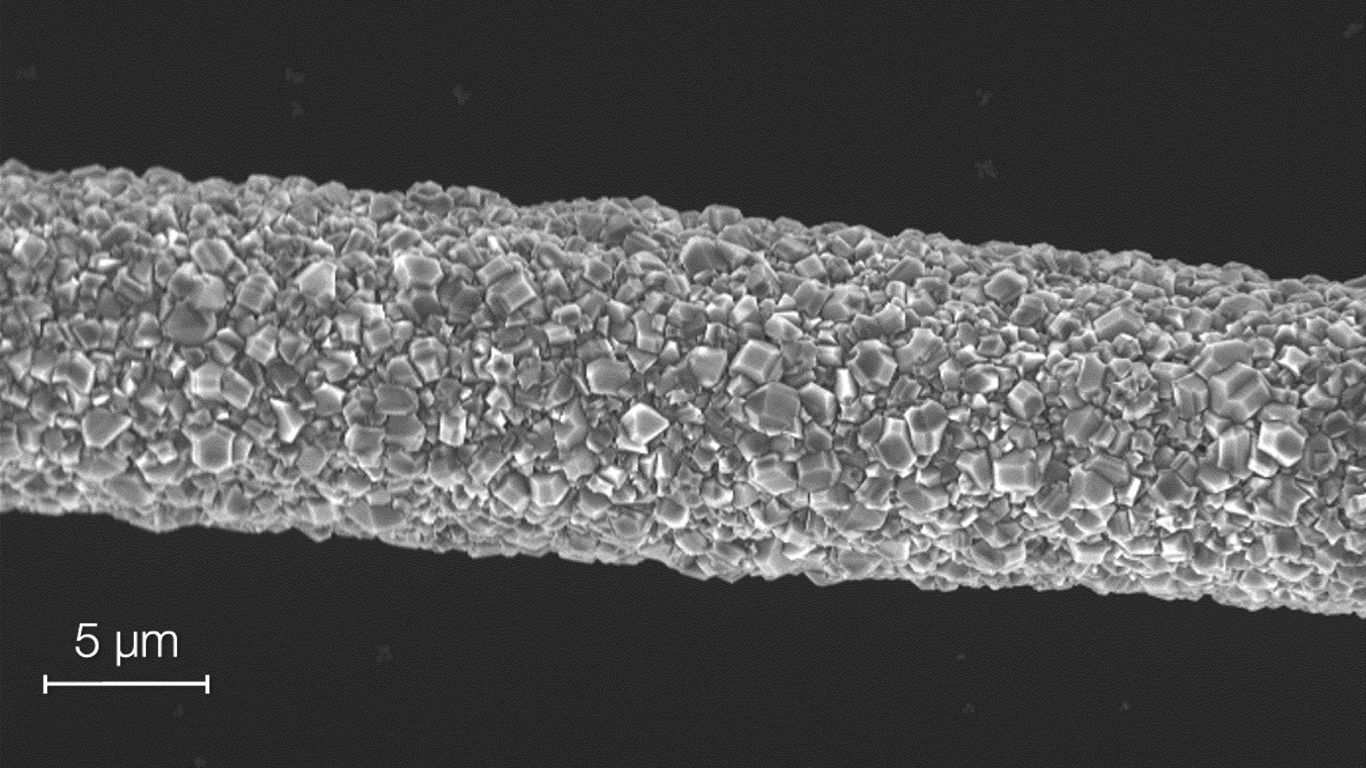
Wenlong and a team of researchers from Monash University have been making a range of flexible sensors by coating elastic materials with interwoven networks of conductive ultra-thin gold nanowires (UGNWs). According to Wenlong, the UGNWs are the world’s thinnest gold nanowires at only 2nm wide — a DNA double helix, by comparison, is just over 2nm in width.
The elastic substrates that the UGNWs are grafted onto are flexible and biologically safe, making them ideal candidates for wearable electronics.
Researchers worked at MCN to create wearable pressure sensors, skin-attachable strain sensors, flexible transparent electrodes and stretchable supercapacitors.
To create a sensor, UGNWs are grown via a solvent-based chemical process that yields a host of hair- like ultra-thin fibres. Once in a liquid suspension, the wires are “painted” onto the elastic material, forming
an interwoven mesh of electrically conductive fibres once the paint dries. When the UGNW-coated elastic
is stretched, the device’s electrical resistance changes too, providing
a real-time and reversible electrical representation of the sensor’s state.
Wenlong exploits these changing properties to detect and monitor movement with an incredibly high degree of precision. His devices offer touch sensitivity and can pick up the tiny forces associated with artery wrist pulses.
“They are able to resolve pressing, stretching, bending, and torsion
forces as well as acoustic vibrations,” Wenlong said. The team’s devices are cheap to manufacture and attachable to almost any surface, allowing accurate measuring of skin or muscle deformation anytime, anywhere.
In the health sector, for example, increased sensitivity of these devices could reveal previously undetected heart defects or be used to monitor detailed hand movements in robotic infrastructure such as advanced prosthetics.
Targeting tumours
At present, the vast majority of cancer treatments work by attacking cancer cells but often affect healthy tissue in the process. The approach, whilst effective, produces a large number of side effects including irreparable and extensive tissue damage, hair loss and nausea, among others.
There has therefore been a great deal of effort devoted to improving the selectivity of cancer therapies in order to minimise collateral damage. Increasingly, the idea is to hunt with a sniper rifle, rather than a shotgun.
Wenlong and his colleagues have gone a step further and developed what is effectively a guided missile that exclusively binds to target tumours before heating defective tissue to the point of destruction. Advancing a treatment called photothermal therapy, the method uses gold nanorods (GNRs) – shorter, wider versions of gold nanowires – which rapidly heat up to 45°C when irradiated by near-infrared (NIR) light.
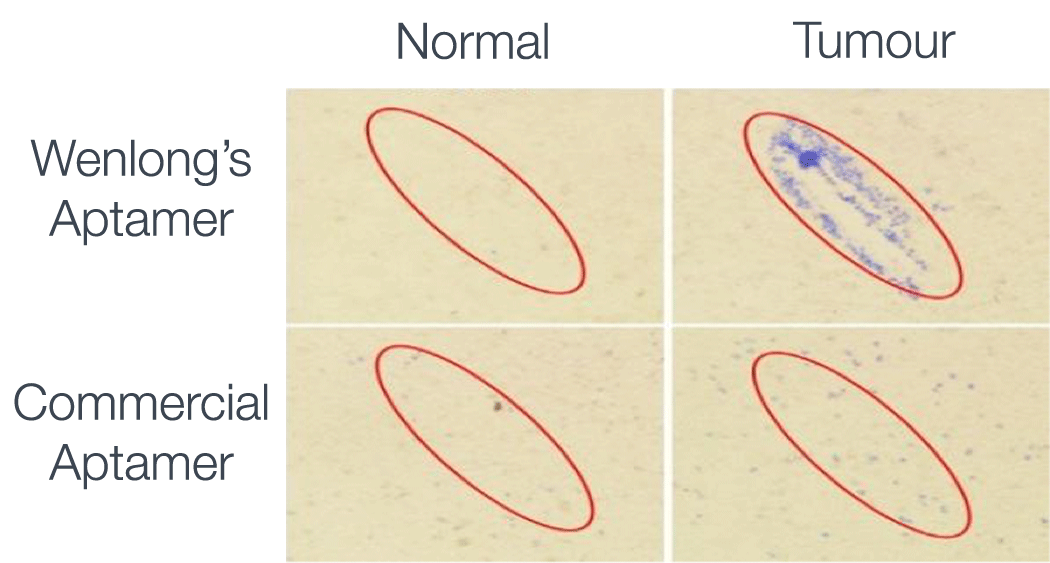
To specifically target the cancerous cells, the GNRs are coated with DNA-specific aptamers that Wenlong selected to identify DNA unique to
the tumour cells. Aptamers are single strands of DNA that bind selectively
to a complementary piece of genetic material. The theory is that once the aptamer binds to the target tumour cell, the GNR is heated with the NIR light and the cell is destroyed.
Wenlong and his team collaborated with a team led by Professor David Jans, NMHRC Senior Principal Research Fellow at Monash University to test the GNR-aptamer “missile” on breast ductal carcinoma cells residing in healthy tissue.
Wenlong’s team was able to observe the selectiveness and efficacy of this process using the MCN’s hyperspectral imaging capabilities. The initial results indicate that once the GNRs had been heated by NIR light, 96 per cent of tumour cells had been successfully targeted and 71 per cent of them were destroyed, with less than one per cent of healthy tissue affected.